Feb 23, 2016
Copenhagen, Denmark – 23 February 2016. Monsenso, a Copenhagen-based technology company, announced today that the Zealand Healthcare Region in Denmark will use the Monsenso mHealth solution for remote patient monitoring of patients with Borderline Personality Disorder (BPD).
The objective of the pilot study is to identify and track the triggers and early warning signs of patients with borderline, as well as to gather clinical evidence with the Monsenso mHealth solution.
The pilot study will be done in collaboration with the Produktion, Forskning and Innovation Centre of the Zealand Healthcare Region in Denmark. This branch focuses on identifying pioneering solutions that have the potential to improve the productivity of hospitals and the quality of life of its patients.
Thomas Lethenborg, CEO at Monsenso ApS, said that the Zealand Healthcare Region will conduct a 20-week pilot study of 30 patients starting in March.
“The Innovation Centre of the Zealand Healthcare Region in Denmark aims to offer its patients high-quality treatment with the latest technology available. With the implementation of the Monsenso mHealth solution, clinicians will be able to access a patient’s historical aggregated data, which will help them offer a better treatment based on evidence,” said Mr Lethenborg.
Jesper Lackman, Project Manager at The Produktion, Forskning and Innovation Centre, said the pilot study would allow them to investigate the benefits of the mHealth solution to improve patient engagement.
“The Produktion, Forskning and Innovation Centre has a strong commitment towards providing patient-centred care. We are continuously looking to improve our patients’ experience and this technology will help us learn more about our patients and their behavioural patterns. Our aim is to use this technology to support the treatment we currently provide to our patients,” said Mr Lackman.
For additional information contact:
Jennifer Highland
Marketing and Communications Manager
Monsenso
+45 81 71 7713
highland@monsenso.com
Jesper Lakman
Innovation Facilitator
PFI – Production, Science & Innovation
+45 8171 2900
jelak@Sundhedsinnovation.com
You can download this article as PDF in English
Feb 16, 2016
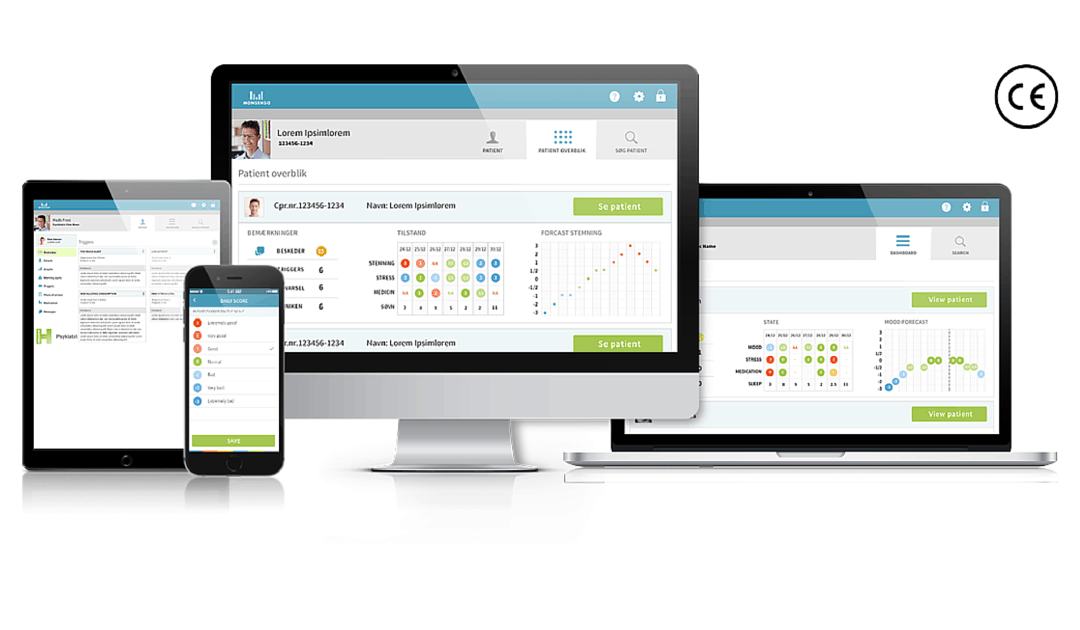
Copenhagen, Denmark – 16 February 2016. Monsenso ApS, a technology leader in mobile health solutions for mental health, today announced that last Friday, its mHealth solution received a CE Mark. The double-loop treatment platform that supports the treatment of major psychiatric disorders received a Class 1 Medical Device Certification under the EU Medical Devices Directive.
The certification has been obtained due to the support received from the Market Development Fund (MMF) in May 2015, which granted 2.9 million to DKK for Monsenso and its partners to obtain the necessary certifications for the solution to be ready for an international launch.
With regards to obtaining the CE Marking Certification, Thomas Lethenborg, CEO at Monsenso, commented “Obtaining this certification is an important milestone for Monsenso since our mHealth solution is based on solid research and has already been clinically validated. Our objective with this certification is to be regarded as a medical application rather than just another app.”
A CE Marking Certification guarantees that a manufacturer’s product meets the essential requirements of all relevant European Medical Device Directives.
“Holding a CE Marking Certification and offering a high level of data security is a demand from our customers across the globe. Therefore, it is important that Monsenso can distinguish itself from all other apps with no clinical evidence or inadequate security measures” added Mr. Lethenborg.
The next step for Monsenso is to obtain the ISO 27001 Data Security Certification, the ISO 13485 Certification, and to become HIPAA and FDA compliant, which is also part of the support granted by the MMF funding.
For additional information contact:
Jennifer Highland
Marketing and Communications Manager
Monsenso
+45 81 71 7713
highland@monsenso.com
You can download this article as PDF in English and Danish
Feb 9, 2016
Copenhagen, Denmark – 9 February 2016. Monsenso ApS, a Copenhagen-based technology company, has been granted 3.065 million DKK by the Innovation Fund to develop further develop their mHealth solution that aims to reduce hospital readmissions of patients with depression and bipolar disorder.
The RADMIS research project, which has been granted a budget of 10.1 million DKK by the Innovation Fund, is being done in collaboration with the Mental Health Services of the Capital Region of Denmark (RHP) and the Danish Technical University (DTU).
Depression and bipolar disorder are the leading cause of disability worldwide, imposing a very high burden for society in terms of cost, lost productivity, and mortality. Besides, patients with affective disorders are hospitalized more frequently than any other patient group. The treatment of depression and bipolar disorder includes medication, psycho-education, and Cognitive Behavioral Therapy (CBT).
The RADMIS research project aims to establish clinical evidence that the Monsenso mHealth solution can reduce the number of hospital readmissions of patients with depression and bipolar disorder. It also seeks to decrease the symptoms and improve the quality of life of outpatients with depression or bipolar disorder receiving smartphone-based CBT.
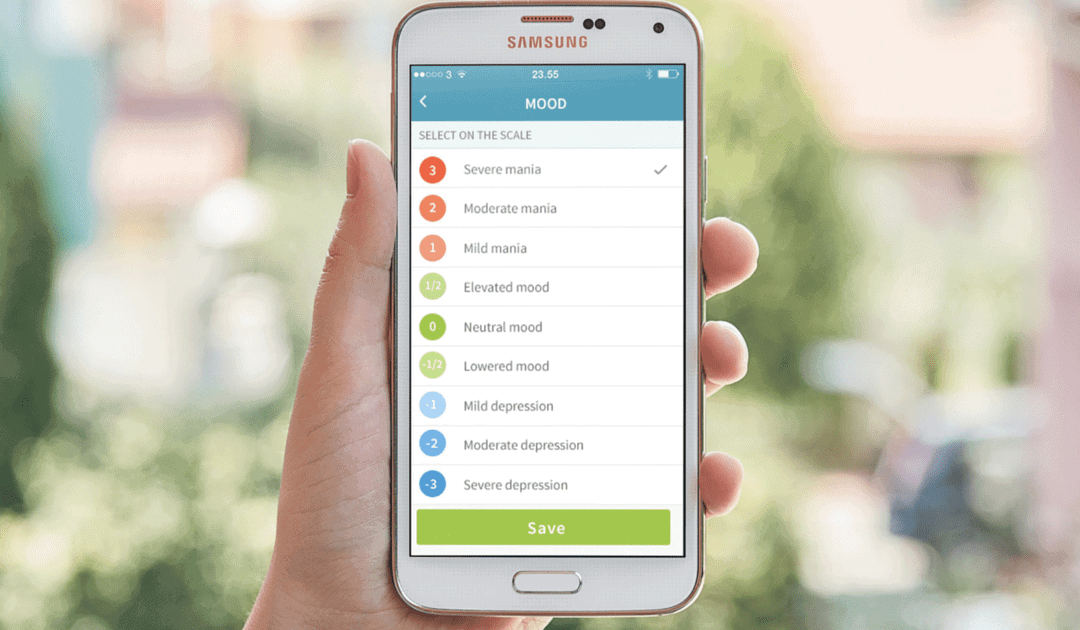
Since smartphones are equipped with powerful sensing, computation and communication capabilities, they provide a unique platform to monitor and treat patients with depression and bipolar disorder. Moreover, smartphones also provide a rich multi-media and communication platform for Ecological Momentary Intervention (EMI) enabling patients to receive context-aware treatment in real-time, including medication reminders and CBT.
The innovation goals of the RADMIS research project are to:
- Research, design, and develop an intelligent smartphone-based monitoring and treatment platform for affective disorders
- Provide clinical evidence to reduce the number and duration of hospital re-admissions of patients with depression and bipolar disorder using smartphone-based treatment
- Provide clinical evidence that smartphone-based CBT improves the health outcomes for patients with depression and bipolar disorder
With regards to the new capabilities that will be added to the Monsenso mHealth solution, Thomas Lethenborg, CEO at Monsenso commented, “This intelligent smartphone-based monitoring and treatment platform for depression and bipolar disorders is the first of its kind; there are no similar solutions available on the market.”
“Once the clinical trial is completed, we will have the necessary evidence to prove that smartphone-based CBT helps reduce hospital readmissions of psychiatric patients. Besides, it allows health care systems to reach out to the 75% of the population in Europe who need treatment, but remain untreated,” added Mr. Lethenborg.
For additional information contact:
Jennifer Highland
Marketing and Communications Manager
Monsenso
+45 81 71 7713
highland@monsenso.comYou can download this article as PDF in English and Danish

Jan 27, 2016
Copenhagen, Denmark – 27 January 2015. Monsenso, a Copenhagen-based technology company, announced today that UK-based, St Andrew’s Healthcare, will implement a new mHealth solution for borderline and depression patients. The mHealth solution, designed and developed by Monsenso, will be used to support the person-centred treatment of inpatients.
Thomas Lethenborg, CEO at Monsenso ApS, said that St Andrew’s will conduct a six-month service evaluation feasibility study to determine if the mHealth solution for borderline and depression patients should also be used to help treat other mental health illnesses.
“St Andrew’s Healthcare has a commitment to providing the best inpatient and outpatient mental health services in the UK. With the implementation of the Monsenso mHealth solution, clinicians will have access to patient’s historical aggregated data that will help clinical staff in the treatment of complex mental health illnesses,” Mr Lethenborg added.
St Andrew’s Healthcare Director of Nursing and Quality, Lesley Boswell said the trial would allow them to investigate how real-time mood monitoring could be used to enhance patient care.
“We are continuously looking to improve our understanding of patient experiences and this technology will help us to access previously hidden mood signals. With a bit of configuration, our aim is to use the technology to help support the therapies and treatment we currently provide to our patients, with views to expanding the programme to other care pathways in the future,” said Ms Boswell.
The Monsenso mHealth solution for borderline and depression is based on a double-loop treatment model. On one side, patients use a smartphone to fill in routine self-assessments that reveal their current state of mind and collect sensor data. The smartphone app also works as a self-management tool that allows patients to manage their symptoms and the behaviours that trigger those symptoms.
On the other side, clinicians use a web portal where they can access all the data collected by the patients’ smartphones anytime, anywhere. The web portal enables clinicians to view relevant information related to each patient.
With more than 4000 employees, St Andrew’s Healthcare delivers effective treatment, therapies, clinical consultancy, vocational rehabilitation and outpatient care across its four sites. The charity provides specialist and secure care pathways in mental health and neuropsychiatry and provides care for young people, adults, and seniors.
For additional information contact:
Jennifer Highland
Marketing and Communications Manager
Monsenso
+45 81 71 7713
highland@monsenso.com
Bonnie Sleeman
Communications Coordinator
St Andrew’s Healthcare
+44 1604 872946
bsleeman@standrew.co.uk You can download this article as PDF in English and Danish
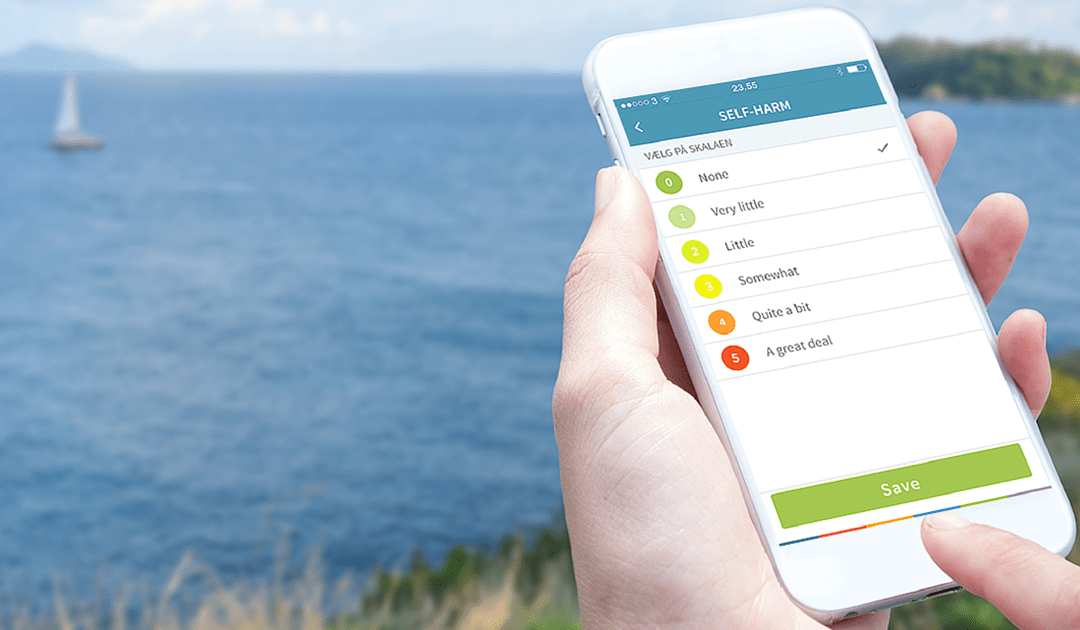
Jan 25, 2016
Copenhagen, Denmark – 25 January 2016. Monsenso ApS, a Copenhagen-based technology company, has been granted 1.575 million DKK to continue the research and development of the existing data-driven, cloud-based, mHealth solution for anxiety and borderline treatment.
The ENTER project (previously called E-Mental project), which has been granted an overall budget of 19 million DKK, is being done in collaboration with The Mental Health Services in the Region of Southern Denmark (MHS), the South Denmark University, Aalborg University, Context Consulting and Monsenso. Monsenso will be involved in two studies for the E-mental project:
The Anxiety Monitoring Study
The objective of the study is to implement and validate the Monsenso mHealth solution for anxiety disorders.
Mobile monitoring solutions can be useful in assisting patients in using skills learned during treatment in real life situations, as well as to monitor and give feedback on progress or lack of progress during therapy. This feedback leads to better outcomes for patients not responding well to treatment.
The mDIARY Study for patients with borderline personality disorder
The objective of the study is to prove that the Monsenso DBT (Dialectical Behavioural Therapy) solution is a reliable and clinically valid way to collect patient data, helping individuals learn these skills at home, speeding up their recovery.
The study will include a three-month pilot study where a randomized percentage of the patients will use either paper-based diary cards or the Monsenso mHealth app. Additionally, the study will also include a two-year randomized controlled trial. During this part of the study, half of the patients will be given an mHealth app that includes coaching suggestions while the second half will use paper-based self-assessments. The number of missing data will be compared, and data quality of the app versus established clinical measures will be evaluated. User satisfaction will be assessed with focus group interviews.
Additionally, a cost-effectiveness study will run in parallel to the main research. The objective of this study is to estimate the economic advantages of implementing the proposed e-mental health solution in the healthcare system.
E-mental health solutions have the potential to reduce the cost of treatment and to enable a larger number of people to be treated. The economic aspects should be adequately investigated with regards to cost-effectiveness as well as financial aspects of implementation in clinical practice. The study aims to determine the cost-effectiveness of Internet interventions compared to the most cost-effective treatment currently available.
With regards to the support obtained from the Innovation Fund, Thomas Lethenborg, CEO at Monsenso commented, “The mHealth solution developed by Monsenso has the potential to revolutionize the treatment of psychiatric illnesses. The support received from the Innovation Fund will provide us with the necessary evidence to validate our solution clinically and financially for the treatment of mental illnesses.”
To learn more click on this link:
For additional information contact:
Jennifer Highland
Marketing and Communications Manager
Monsenso
+45 81 71 7713
highland@monsenso.com You can download this article as PDF in English

Dec 1, 2015
Copenhagen, Denmark – 1 December 2015. Monsenso Aps, an innovation leader in mobile health (mHealth) solutions for mental health, announced today the acquisition of DayBuilder Solutions, a local competitor developing an mHealth solution for depression to strengthen focus on mHealth for mental health.
The acquisition will consolidate Monsenso in the Danish market and strengthen their focus internationally. Starting from today, Monsenso will take over DayBuilder’s obligations to customers and researchers. Besides acquiring new customers with this merger, Monsenso will also benefit from additional research and technology that will support the future development of leading mobile health solutions for mental health.
With regards to the merger, Thomas Lethenborg, CEO at Monsenso, commented, “With this merger, two evolving Danish companies can now present a unified front locally and internationally, rather than being competitors. This merger has made us a stronger player in the development space of mHealth solutions; therefore, clinicians and patients will benefit from this outcome.”
“I believe that Monsenso has the capability to dominate the market nationally and internationally, and with our joint efforts we have the potential to help thousands of people suffering from mental illness worldwide,” commented Philip Kaare Løventoft, CEO at DayBuilder Solutions.
After the merger is completed, Monsenso will take over a new research project called SAFE2, which was handled by DayBuilder Solutions in collaboration with Rigshospitalet, the University Hospital in Denmark.
Currently, the Monsenso mHealth solution is being used by the different health care regions in Denmark to support the treatment of patients suffering from schizophrenia, major depressive disorder(MDD), post-traumatic stress disorder (PTSD), anxiety, and borderline personality disorder.
For additional information contact:
Jennifer Highland
Marketing and Communications Manager
Monsenso
+45 81 71 7713
highland@monsenso.com You can download this article as PDF in English and Danish