Life science
REAL-WORLD DATA
COLLECTION
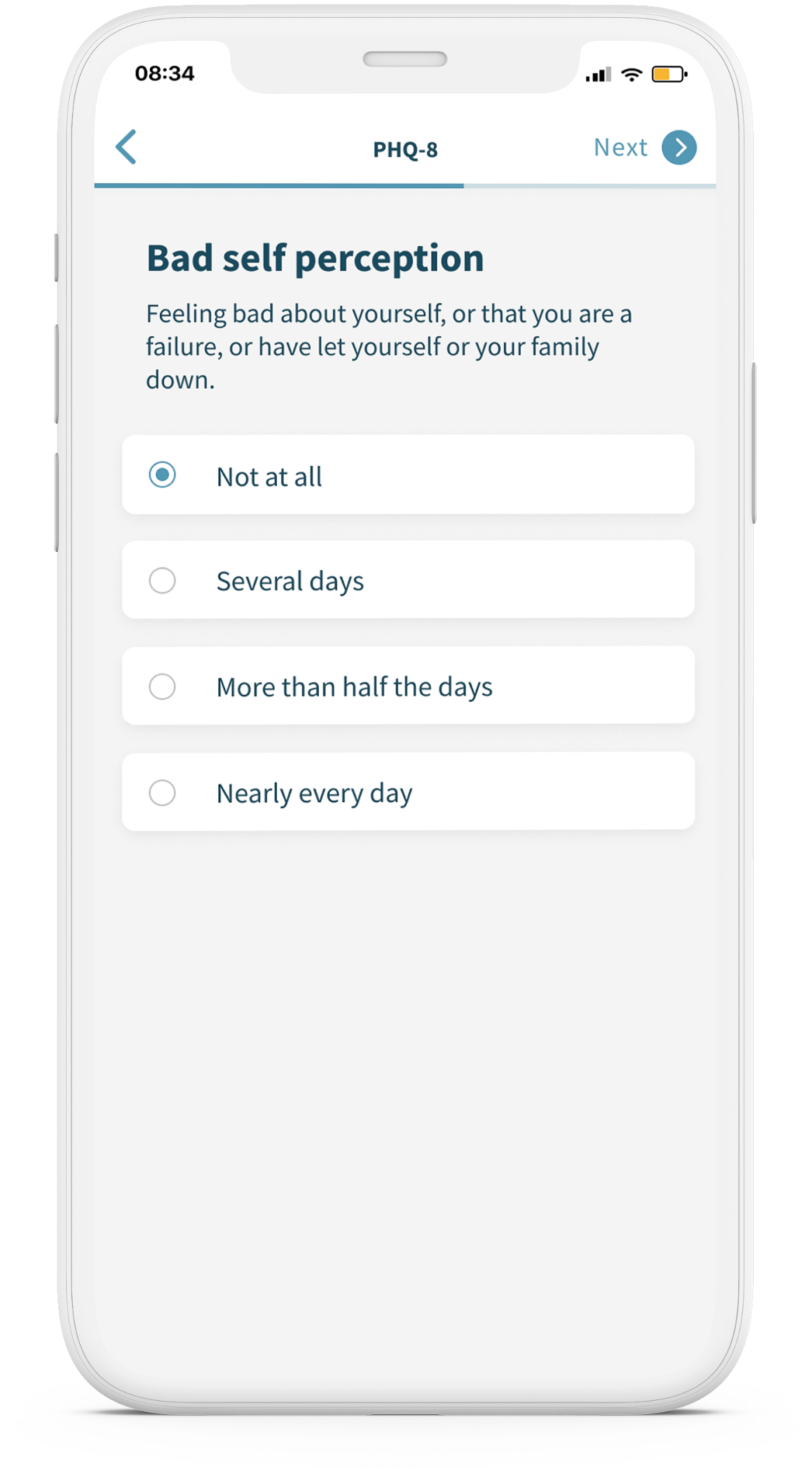
Monsenso enables the remote collection of patient-
generated data and device-generated data from
clinical, post-approval to real-world studies.


POWERING REAL-WORLD EVIDENCE STUDIES
The Monsenso solution empowers CROs and pharmaceutical companies to capture real-world data, revealing patient insights that could enhance a trial’s efficacy and speed up the drug development process.
Our agile, evidence-based solution enables you to remotely assess all your subjects 24/7 across the product life cycle.
Our digital health solution enables the remote collection of patient-generated data and device-generated data from clinical, post-approval to real-world studies.
Monsenso is a CE Class 1 medical device that securely captures patient-generated data using ePRO and a suite of sensors and wearables to provide a more holistic view of patients’ journey.
Real-World Evidence generation
Monsenso enables real-world evidence (RWE) generation through patient-generated & device-generated data collection across the product life cycle.
Clinical Development
Enable patient-centred drug development with our solution to impact outcomes that matter to patients. Our evidence-based solution is developed with and for patients with mental health and psychiatric conditions to reduce their burden of contributing to research.
Post-Market Studies
Equip global, multi-centre, non-interventional, longitudinal studies with our solutions to measure safety and efficacy, describe patient, drug use, and clinical development across treatment pathways in real-world medical and clinical practice for regulators.
Companion Products
Engage patients with our solutions to enable personalised care, education, self-management, behavioral change, symptom and disease progression monitoring, and medication adherence. Demonstrate value to patients, prescribing physicians and payers.
Digital Biomarkers, Digital Therapeutics & Personalised Medicine
Enhance the development and validation of digital endpoints, biomarkers, therapeutics, and pharmacogenomics testing in the real-world setting using our capabilities for continuous stream of active and passive patient data capture.
BENEFITS OF WORKING WITH US

Therapeutic expertise
7 years of clinical experience within mental health and psychiatric disorders, amongst others: depression, bipolar disorder, anxiety, schizophrenia, borderline personality disorder, addiction and substance abuse.

Track record
Extensive track record in global deployment within academia, healthcare, social care and pharmaceutical companies across a variety of mental health disorders.

Scalable for fast deployment
A configurable, customisable cloud-based/SaaS solution, that can be quickly configured to meet the needs of the target patient population and care pathway.

Regulatory, data privacy & security
Protocols designed to meet regulatory requirements. CE-marked as Class 1 Medical Device, Cyber Essentials certified and ISO 13485 and ISO 27001 compliant.
REQUEST A DEMO
Our expert team is ready to demonstrate the Monsenso real-world data collection solution and to answer any questions you might have.

References
[1] FDA: Https://www.fda.gov/science-research/science-and-research-special-topics/real-world-evidence
[2] Roberts, M.H., Ferguson, G.T. (2021). Real-World Evidence: Bridging Gaps in Evidence to Guide Payer Decisions. PharmacoEconomics Open 5, 3–11. https://doi.org/10.1007/s41669-020-00221-y
[3] Calvert, M.J., O’Connor, D.J., & Basch, E.M. (2019). Harnessing the patient voice in real-world evidence: the essential role of patient-reported outcomes. Nature Reviews Drug Discovery, 18(10), 731-732.
[4] Bourke, A., Dixon, W.G., Roddam, A., et al (2020). Incorporating patient-generated health data into pharmacoepidemiological research. Pharmacoepidemiology and Drug Safety, (12):1540-1549. DOI: 10.1002/pds.5169.
[5] Semahegn, A., Torpey, K., Manu, A. et al. (2020) Psychotropic medication non-adherence and its associated factors among patients with major psychiatric disorders: a systematic review and meta-analysis. Systems Reviews 9, 17 . https://doi.org/10.1186/s13643-020-1274-3
[6] Sun SX, Liu GG, Christensen DB, Fu AZ. Review and analysis of hospitalization costs associated with antipsychotic nonadherence in the treatment of schizophrenia in the United States (2007). Current Medical Research Opinion.(10):2305-12. doi: 10.1185/030079907X226050. PMID: 17697454.


When you have a mental illness you need to pay attention to your lifestyle in order to have a good life. Monsenso is very helpful, it reminds me to take my medication and provides me with an overview of my triggers and warning signs”
Patient with bipolar disorder