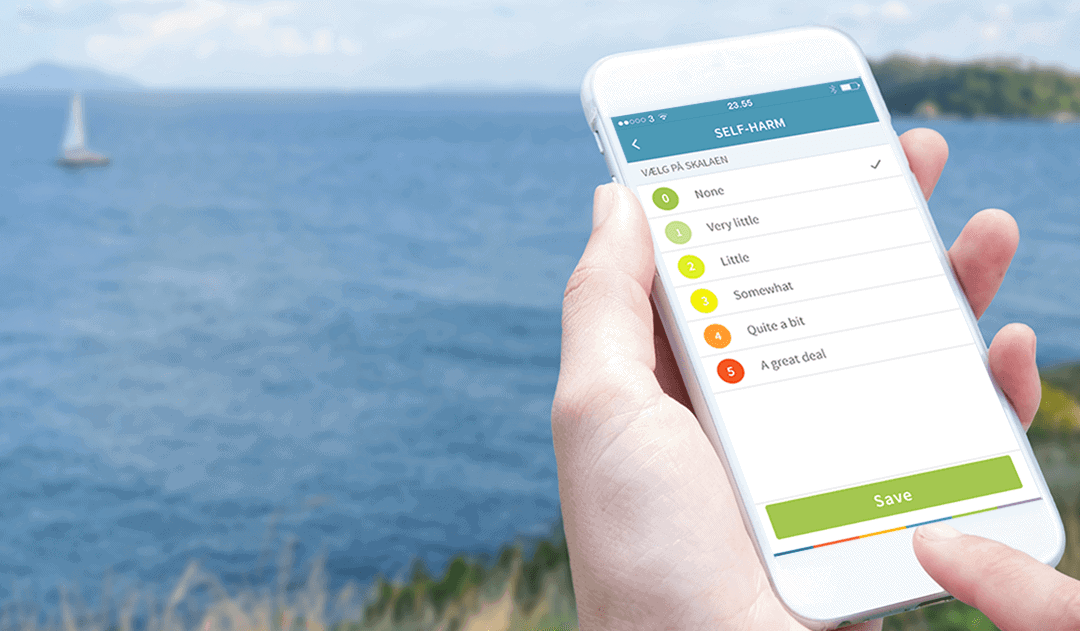
Smartphone-based DBT
The objective of the mDiary Study is to evaluate the Monsenso mHealth solution for borderline patients with respect to reliability, validity and clinical efficacy as an adjunct to DBT.
Borderline is a severe mental disorder characterised by difficulties with regulating emotions, leading to unstable and self-destructive behaviours. Borderline is difficult to treat, especially when co-occurring with other disorders. DBT is the best validated treatment for borderline, showing an improved health outcome compared to other types of treatment.
DBT uses skills training and self-monitoring as the pillar of treatment, teaching patients to regulate their emotions and reduce problem behaviour. Traditionally, self-monitoring was made through paper-based self-assessments. However, new technology has generated an alternative to ecological momentary assessments of problematic behaviours with the additional functionality of prompting patients to practice skills targeting emotion regulation.
Methods
Participants will be borderline patients from psychiatric DBT units. The study includes two phases: the first phase will be a three-month pilot study where patients will be randomised to use either the pen and paper version of the diary card or the Monsenso mHealth app. The number of missing data will be compared, and data quality of the app versus established clinical measures will be evaluated. User satisfaction will be assessed with focus group interviews.
The second phase will include a two-year multi-centre randomised controlled trial. In both conditions patients will be followed for one year. Half of the patients will receive the mHealth app that includes mobile DBT (mDBT) coaching suggestions and instructions on how to use it. The second half will be asked to download an app that only collects sensor data, but will continue to use the traditional paper-based self-assessments.
Expected results
The mDiary Study will prove that the Monsenso smartphone-based DBT is a reliable and valid way to collect patient-data, helping individuals learn these skills at home, speeding up their recovery.
To read the press release click here.